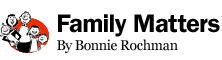Adam Foye, 11, and his mother Sarah have been trying to find out what is causing his muscle weakness
The mystery is over. Doctors now know what’s ailing Adam Foye.
For Adam’s entire life (read his story here), doctors have been trying to figure out why his muscles are so weak that he can’t carry his sixth-grade schoolbooks or walk down a grocery aisle without getting exhausted.
And now the Pine Brook, N.J., boy and his family have their answer. Researchers who sequenced his genome have zeroed in on what they believe is the culprit: changes in a gene called titin. “We are pretty thrilled,” says Sarah Foye, Adam’s mom. “We’ve been waiting 11 years for this.”
(MORE: Will My Son Develop Cancer? The Promise (and Pitfalls) of Sequencing Children’s Genomes)
The Foye family learned the results as part of a contest sponsored by Boston Children’s Hospital. The competition — called CLARITY, for Children’s Leadership Award for the Reliable Interpretation and Appropriate Transmission of Your Genomic Information — challenged 23 teams of researchers from across the globe to interpret the genomes and exomes of three families with children who suffer from undiagnosed diseases.
Whole-genome sequencing analyzes a person’s entire genetic code, while exome sequencing looks just at the part that codes for proteins. As sequencing moves beyond the research lab and into mainstream medicine, the hospital launched the contest to help streamline “best practices” and develop ways to clearly interpret and communicate test results to help families like the Foyes.
(MORE: Decoding Cancer: Scientists Release 520 Tumor Genomes from Pediatric Patients)
Winners — those groups that were best able to pinpoint the children’s genetic mutations and explain them in a way that makes sense to patients and their doctors — were announced Wednesday in San Francisco at the annual meeting of the American Society of Human Genetics.
First prize went to the Division of Clinical Genetics at Brigham and Women’s Hospital in Boston, which received $15,000 for what judges said was the best blend of “analysis, clarity and utility” in the reports it generated for the three families. A teams from the University of Iowa — which was lauded for taking a unique approach in returning unanticipated genetic results — and a German team that was the only group to accurately identify every potential genetic mutation in all of the families, each received $5,000. Teams from Slovenia, Ohio, Sweden, San Diego and Pennsylvania also earned special mentions.
Being able to home in on the cause of Adam’s muscle weakness — a disease called centronuclear myopathy — helps the Foyes and Adam’s physicians better understand the protein that’s impaired. That, in turn, gives them insight into the mechanisms responsible for his particular form of the disease, which will enable them to target specific medications and avoid using ones to which he won’t respond.
(MORE: Why Cheaper Genetic Testing Could Cost Us a Fortune)
The discovery also illustrates exactly how genome sequencing — which in this case compares Adam’s genome with that of someone unaffected by his disease in order to isolate the genes responsible — can prove so useful, by quickly teasing out potentially relevant sections of DNA that could become targets for new drugs.
But as exciting as the finding is, it’s hardly the end of the Foyes’ medical quest.
“Until you find the part in the entire machine that is altered or broken, you don’t have any reasonable hope of trying to fix it,” says Dr. David Margulies, executive director of the Gene Partnership at Boston Children’s, which seeks to apply genomics and genetics to research and clinical medicine. “Once you’ve identified the part that is altered, you don’t have any assurance that you can fix it, but at least you have a starting point.”
That’s exactly how the Foyes feel. “As my husband says, this is not our final destination on our medical journey, but it’s an important milestone,” says Sarah Foye, who traveled to San Francisco to hear the results in person. “Now we want to work toward a treatment.”
Adam, who was told of the results, has been asking questions about titin. Foye overheard him talking about the discovery with one of his friends. “I heard him say, I want to be able to run faster than you,” she says. “I don’t know if that will ever happen, but I would really love that.”